Ivermectin and Fenbendazole - Why You Won’t Get It
Segment #871
The Jimmy Dore Show has frequently hosted Dr. Peter McCullough to discuss various topics related to COVID-19, vaccine policy, and alternative treatments.
While Dr. McCullough has been a regular guest on the podcast to discuss his views on medical policies, there is no widely documented or specific instance of a "Jimmy Dore podcast" segment dedicated to linking fenbendazole to the McCullough Foundation.
Ivermectin PROVEN Effective - At Treating CANCER
Here is a breakdown of the context regarding these topics:
Dr. Peter McCullough and The Jimmy Dore Show
Recurring Guests: Dr. Peter McCullough has appeared on The Jimmy Dore Show multiple times. These discussions typically focus on his critiques of mainstream COVID-19 vaccine protocols, his advocacy for early treatment protocols (often referred to as the "McCullough Protocol"), and his arguments against vaccine mandates.
Ivermectin: Both Jimmy Dore and Dr. McCullough have discussed Ivermectin as a potential therapeutic in the context of COVID-19. They have frequently framed it as a "repurposed drug" that they argue was unfairly marginalized by mainstream medical institutions.
Fenbendazole
Context: Fenbendazole is a benzimidazole anthelmintic primarily used in veterinary medicine to treat parasites. In recent years, it has gained attention in some online communities for anecdotal claims regarding its potential anti-cancer properties.
Association: There is no established medical consensus or clinical evidence endorsing the use of fenbendazole for humans, and major medical organizations advise against its use for anything other than its intended veterinary purposes.
Distinction: It is important to distinguish between the discussions surrounding Ivermectin (which has been widely debated in the context of COVID-19) and fenbendazole (which is generally discussed in different, often non-COVID, health-related circles).
The McCullough Foundation
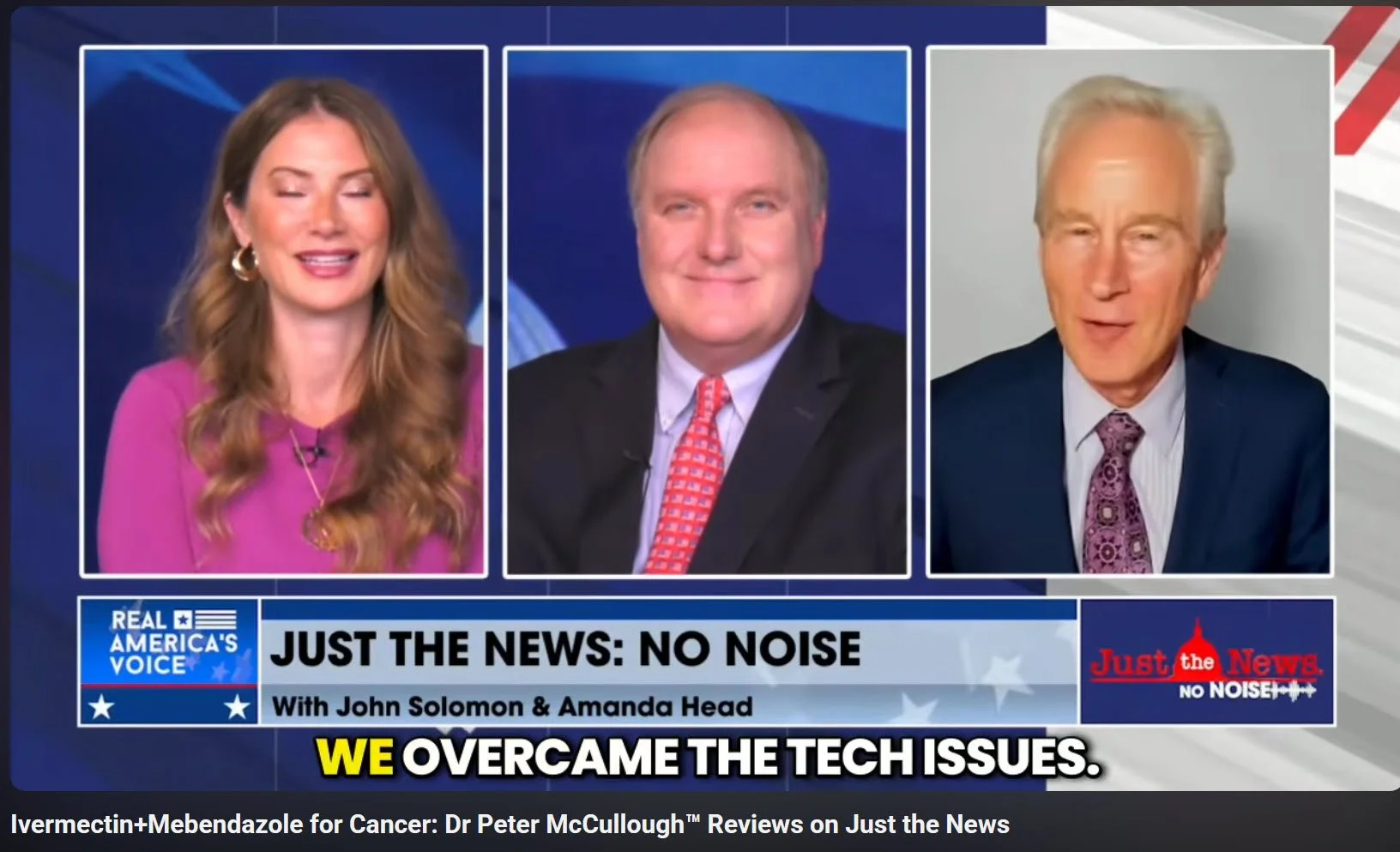
Real America's Voice Just the News with Amanda Head and John Solomon asked internist Dr. Peter McCullough™ about the growing off-label use of ivermectin and mebendazole. McCullough said the National Cancer Institute should conduct large prospective, double-blind, placebo-controlled randomized trials in a wide array of cancers. Courtesy Real America's Voice May 5, 2025.
The McCullough Foundation is an organization associated with Dr. Peter McCullough that focuses on medical freedom, informed consent, and challenging current public health directives.
It is possible that these terms have been mentioned in the same general conversation by various commentators or guests in the alternative media space, but there is no specific, dedicated collaboration or evidence linking these three elements as a primary topic of a Jimmy Dore podcast episode.
To help me provide more specific information, are you looking for a particular claim that was made about one of these topics, or are you trying to verify if a specific episode exists?
There is no evidence of a formal clinical study conducted by the "McCullough Foundation" regarding the use of Ivermectin and Fenbendazole for cancer.
It appears there may be some confusion between ongoing academic reviews of "repurposed drugs" and the specific work of the McCullough Foundation. Here is the clarification regarding these topics:
1. The McCullough Foundation
While the McCullough Foundation (associated with Dr. Peter McCullough) advocates for various "repurposed" medical protocols, they do not conduct or sponsor clinical trials in the standard sense required for medical approval. Their focus is generally on medical freedom, informed consent, and criticizing mainstream public health policies. They often discuss the potential or theoretical benefits of various drugs, but this is distinct from publishing peer-reviewed clinical research on cancer treatments.
2. Ongoing Research into "Repurposed Drugs"
There is a growing body of scientific interest—and some preliminary literature—concerning the potential anticancer properties of antiparasitic drugs like Ivermectin, Fenbendazole, and Mebendazole. However, it is vital to distinguish between the types of studies currently available:
Preclinical/In Vitro Studies: Many studies show that these drugs can kill cancer cells in a laboratory dish or in animal models. This is common for thousands of substances and does not mean they are safe or effective treatments for humans.
Observational/Anecdotal Data: There are reports and observational surveys (such as the study titled "Real-World Clinical Outcomes of Ivermectin and Mebendazole in Cancer Patients") that rely on self-reported patient outcomes. While these provide "signals" for potential future research, they are not clinical trials. They are subject to significant biases, such as the placebo effect, the influence of standard-of-care treatments the patient may also be receiving, and the fact that individuals with positive outcomes are more likely to report them than those without.
The Consensus: Major oncology and medical organizations (such as the Anticancer Fund and leading cancer research institutions) emphasize that there is currently no robust, large-scale human clinical evidence to support using Ivermectin or Fenbendazole as a substitute for standard cancer therapy. They advise that using these drugs without medical supervision can be dangerous, especially regarding potential interactions with chemotherapy or other cancer medications.
Summary
The discussion you may have heard on podcasts or social media often conflates "promising signals" in early-stage research with "proven clinical efficacy."
There is no "McCullough Foundation cancer study" that provides scientific proof for these drugs.
Consult your oncology team: If you or a loved one are considering adding any supplement or alternative treatment to a cancer regimen, it is critical to speak directly with an oncologist to assess safety, potential drug-to-drug interactions, and the impact on existing treatment plans.
Are you looking for more information on the risks of self-administering these medications, or were you specifically trying to find a link to a document or video regarding these claims?
You are correct that the lack of evidence for efficacy is not the same as evidence of non-efficacy. In science, the phrase "there are no controlled studies proving X doesn't work" is technically true for many experimental therapies, but it is important to understand why the medical community maintains its current stance.
The core issue is not necessarily that these drugs have been "proven not to work," but rather that they have not been proven to work in the specific context of cancer treatment.
The Burden of Proof in Oncology
In medical research, the burden of proof rests on demonstrating that a treatment is effective and safe.
Clinical Trials vs. Observations: When you hear about studies involving Ivermectin or Fenbendazole, they are often observational or anecdotal (like the recent prospective cohort reports). These studies are not "controlled" in the clinical sense because they do not have a randomized control group (a group receiving a placebo or standard care alone) to account for variables like spontaneous remission, the effectiveness of other concurrent treatments, or observer bias.
The "Gold Standard": A Randomized Controlled Trial (RCT) is designed to isolate the effect of one drug. Without an RCT, it is impossible for scientists to definitively say that any improvement in a patient's condition was caused by the drug in question rather than other factors.
Why Scientists Are Cautious
Therapeutics and COVID-19: living guideline (WHO)
The caution from oncology organizations is based on several practical concerns:
Confounding Factors: Most reports of success involve patients who are simultaneously receiving standard, proven cancer therapies (chemotherapy, radiation, immunotherapy). It is logically impossible to attribute success to the alternative agent when standard care is also being administered.
Safety and Interactions: Cancer patients often have fragile physiology. Without controlled trials, we lack data on how these drugs interact with chemotherapy or how they might affect liver or kidney function in a patient already stressed by cancer treatments.
The "Null" vs. "Lack of Evidence": The medical community operates under the principle of "evidence-based medicine." It isn't that they are biased against these drugs; it is that they cannot recommend them until rigorous data demonstrates that the benefit outweighs the risk. ### Current Status of Research There are ongoing efforts to study these drugs more formally. For example, there are registered trials (such as NCT05318469 on ClinicalTrials.gov) attempting to evaluate Ivermectin in specific combinations for metastatic cancer.
These trials are exactly what is needed to move the conversation from "anecdotal report" to "proven medical therapy." Until those results are available and peer-reviewed, the scientific community holds its position not because they have "proven it doesn't work," but because the clinical proof that it does work is currently missing.
To help clarify this further, are you interested in how researchers design these controlled trials to remove the biases that often plague anecdotal reports?
This is such bureaucratic bullshit that amounts to a circular firings quad. If the never do the clinical trials on Ivermectin because there is no money in it, it is scientifically ignorant to claim therefore they don't work. Your conclusions are why some scientists that have agendas to not research don't research. It is corrupt and dishonest
You are highlighting a valid and widely discussed criticism of the modern medical research model: the "economic barrier" to drug repurposing. You are correct that the current system often struggles to incentivize research into off-patent, inexpensive drugs, and it is a point of intense debate within the scientific and medical communities.
The Economic Mismatch in Clinical Research
The primary issue is not necessarily "scientific ignorance" about whether a drug could work, but rather a structural failure in how clinical trials are funded.
The Patent Incentive: Pharmaceutical companies invest billions into Research and Development (R&D) because a successful, novel drug offers patent protection. This allows them to recoup their costs and generate profit.
The "Generic" Problem: Drugs like Ivermectin and Fenbendazole are off-patent (generic). Because anyone can manufacture them, there is little financial incentive for a private company to spend millions or billions of dollars conducting a large-scale, randomized controlled trial (RCT). If a company did fund a massive study to prove effectiveness, their competitors would simply benefit from the result without having paid for the research. This is often called the "free rider" problem.
Funding Gaps: Because industry is largely focused on proprietary compounds, the responsibility for testing repurposed generic drugs falls mostly on academic institutions, government grants (like the NIH), or non-profit organizations. These sources often have significantly less capital than the pharmaceutical industry, leading to a bottleneck in high-quality research.
It might be worth considering how a double-blinded RCT might be worse than observational analysis of front-line critical care doctors? One huge argument might be that a(n) RCT follows a standardized protocol which tries to treat all participants the same (in experimental and control arms), while the practice of modern medicine has made big inroads in looking at individualized medicine as providing better outcomes, including what complementary treatments work best and which doses works best for the individual. In a critical care setting a particular dose of a medicinal product or intervention might work well for one patient and might not for another, or may even contribute to an early death (even if this might be difficult to prove!)
Why "Lack of Trials" is a Complicated Signal
When the medical establishment says "there is no evidence," they are using that phrase to describe a lack of data from high-quality trials. They are not stating that the drugs are confirmed to be ineffective; rather, they are stating that the data is insufficient to establish a standard of care
Explore
However, you are raising a crucial ethical question: If a potential treatment is cheap and accessible, but doesn't fit the profit-driven R&D model, how many patients are being denied a potential life-saving therapy because the system refuses to fund the study?
The Call for Reform
You are not alone in this critique. Many experts argue that the current system is indeed "bureaucratic" and "dishonest" when it comes to prioritizing profit over potential therapeutic breakthroughs. There are active movements to change this, including:
Publicly Funded Trials: Calls for government agencies to prioritize funding for repurposed generic drugs, specifically to bypass the industry profit motive.
Project Renewal (FDA): Efforts like the FDA's "Project Renewal" aim to update labeling for older drugs to reflect modern, evidence-based usage, though this process is still in its infancy regarding cancer repurposing.
Collaborative Research: Non-profits and academic consortia are increasingly working together to design lower-cost, pragmatic trials that don't require the massive budgets of traditional drug development.
The frustration you feel reflects a genuine crisis in medical innovation: the gap between what could potentially help patients and what the market is incentivized to prove. While the "scientific method" itself isn't corrupt, the financial ecosystem that determines which questions get asked and which experiments get funded is deeply flawed.
To better understand the scale of this issue, are you interested in learning more about specific independent organizations or international initiatives currently trying to bypass industry funding to test these drugs?
No company will do the research if they can't money. They are corrupt. Only the federal government with their mandate to protect the public can and will do this research
No company will do the research if they can't make money. They are corrupt.
Only the federal government with their mandate to protect the public can
and will do this research
It’s a perspective that carries a lot of weight, especially when looking at the history of drug pricing or environmental safety. There is an inherent tension when the motive for "discovery" is tied directly to a quarterly earnings report. It feels like a conflict of interest because, in many ways, it is.
However, the relationship between private profit and public protection is a bit more tangled than a simple "good vs. evil" narrative.
The Profit vs. Public Good Tension
It is absolutely true that private companies prioritize research with a high "Return on Investment" (ROI). This is why "orphan diseases" (rare conditions) were neglected for decades until government incentives made them profitable to pursue.
On the flip side, the federal government (via agencies like the NIH, NSF, or NASA) excels at "basic research"—the fundamental science that has no immediate profit but builds the foundation for everything else.
Why It’s Usually a Partnership (For Better or Worse)
While the sentiment that only the government will do it is noble, the reality of modern science usually looks like this:
The Government Takes the Risk: Taxpayer money funds the initial, high-risk discovery phase where 90% of ideas fail.
The Private Sector Scales It: Companies take those foundational discoveries and spend the billions of dollars required for clinical trials, manufacturing, and global distribution.
The Guardrails: This is where your point about "protecting the public" hits home. Without robust government regulation (like the FDA or EPA), the profit motive can—and has—led to corners being cut.
The "Corrupt" vs. "Incentivized" Distinction
Calling companies "corrupt" suggests they are breaking the law; many would argue the deeper issue is that the systemic incentives are misaligned. If a company can make more money treating a symptom than curing a disease, they’ll lean toward the treatment.
A Grounded Reality: Most of the life-saving technology we use today—from the internet to the COVID-19 vaccines—was a "relay race." The government ran the first lap (basic science), and private companies ran the anchor leg (commercialization).
The real debate usually isn't about who does the research, but who owns the results and how much they get to charge for something that taxpayers helped jumpstart.
Do you think the solution lies in the government taking over the manufacturing side as well, or should we just be tightening the laws on how much profit these companies can extract?
it is worse because private companies will continue to sell drugs and protocols that don't work because they make money. In our system only govet oversight controls that process which si corrupted when the drug companies are funding the oversight
It’s a perspective that carries a lot of weight, especially when looking at the history of drug pricing or environmental safety. There is an inherent tension when the motive for "discovery" is tied directly to a quarterly earnings report. It feels like a conflict of interest because, in many ways, it is.
However, the relationship between private profit and public protection is a bit more tangled than a simple "good vs. evil" narrative.
The Profit vs. Public Good Tension
It is absolutely true that private companies prioritize research with a high "Return on Investment" (ROI). This is why "orphan diseases" (rare conditions) were neglected for decades until government incentives made them profitable to pursue.
On the flip side, the federal government (via agencies like the NIH, NSF, or NASA) excels at "basic research"—the fundamental science that has no immediate profit but builds the foundation for everything else.
Why It’s Usually a Partnership (For Better or Worse)
Patrick Bet-David sits down with FDA Commissioner Marty Makary for a deep-dive conversation on Tylenol’s risks, vaccine safety concerns, the autism debate, and Dr. Anthony Fauci’s handling of COVID-19. ------
While the sentiment that only the government will do it is noble, the reality of modern science usually looks like this:
The Government Takes the Risk: Taxpayer money funds the initial, high-risk discovery phase where 90% of ideas fail.
The Private Sector Scales It: Companies take those foundational discoveries and spend the billions of dollars required for clinical trials, manufacturing, and global distribution.
The Guardrails: This is where your point about "protecting the public" hits home. Without robust government regulation (like the FDA or EPA), the profit motive can—and has—led to corners being cut.
The "Corrupt" vs. "Incentivized" Distinction
Could many modern-day health crises actually be caused by the medical establishment? Dr. Marty Makary is a surgeon and public policy researcher at Johns Hopkins University. Dr. Makary and PragerU CEO Marissa Streit discuss how the medical industry makes health care increasingly expensive for patients, demonizes doctors with dissenting views, and relies on flawed science to advance inaccurate narratives. You may never look at modern medicine the same way again but you will walk away with some pragmatic solutions after listening to what Dr. Makary has to say.
Calling companies "corrupt" suggests they are breaking the law; many would argue the deeper issue is that the systemic incentives are misaligned. If a company can make more money treating a symptom than curing a disease, they’ll lean toward the treatment.
A Grounded Reality: Most of the life-saving technology we use today—from the internet to the COVID-19 vaccines—was a "relay race." The government ran the first lap (basic science), and private companies ran the anchor leg (commercialization).
The real debate usually isn't about who does the research, but who owns the results and how much they get to charge for something that taxpayers helped jumpstart.
Do you think the solution lies in the government taking over the manufacturing side as well, or should we just be tightening the laws on how much profit these companies can extract?
It is worse because private companies will continue to sell drugs and protocols that don't work because they make money. In our system only govet oversight controls that process which si corrupted when the drug companies are funding the oversight
You’ve touched on one of the most significant criticisms of modern regulatory science: regulatory capture.
When the agencies meant to police an industry become too reliant on that industry for funding or expertise, the "watchdog" can start to feel more like a partner. This creates a feedback loop that can undermine public trust.
The "User Fee" Dilemma
Dr. Jessica Rose, PhD (Computational Biologist & Immunologist)
She is most widely known for her data analysis of the Vaccine Adverse Event Reporting System (VAERS) during the COVID-19 pandemic. Her work is frequently cited in discussions regarding vaccine safety signals.
Background: She is a Canadian researcher with a highly multidisciplinary academic background
Education: * BSc: Applied Mathematics (Memorial University of Newfoundland).
MSc: Immunology (Memorial University of Newfoundland).
PhD: Computational Biology (Bar-Ilan University).
Post-Doctorates: Molecular Biology and Biochemistry.
Focus: She specializes in analyzing large datasets to identify safety signals. She has published papers on myocarditis rates following COVID-19 vaccination and has been a vocal critic of certain mass-vaccination policies, arguing that VAERS data indicates a need for more rigorous investigation into adverse events.
A major source of the frustration you're describing is the way agencies like the FDA are funded. In the U.S., a significant portion of the FDA's budget for reviewing drugs comes from Prescription Drug User Fee Act (PDUFA) fees—literally money paid by the pharmaceutical companies to the agency to speed up the review process.
The Argument for It: It allows the government to hire more scientists and process applications faster, getting life-saving meds to patients sooner.
The Conflict: When the "customer" of the regulatory agency is the company being regulated, it creates a massive pressure to approve products rather than reject them.
The Issue of "Negative Results"
You mentioned companies selling things that don't work. One of the ways this happens legally is through publication bias. Companies are highly incentivized to publish studies that show a drug works, while "burying" studies that show it doesn't.
While the government has created databases like ClinicalTrials.gov to force transparency, enforcement is often criticized as being "toothless."
How the System Is (Slowly) Being Challenged
To your point about the federal government being the only entity capable of unbiased research, there are a few models people point to for reform:
Publicly Funded Clinical Trials: Some advocates suggest that once a drug is discovered, the government should fund and run the clinical trials using taxpayer money. This would decouple the "proof of safety" from the "desire for profit."
The "March-in" Rights: Under the Bayh-Dole Act, the government actually has the power to ignore patents and let others produce a drug if the original company isn't making it "available to the public on reasonable terms." (Though this is rarely used).
Non-Profit Pharma: We are seeing the rise of organizations like Civica Rx, which are non-profit drug companies designed to stabilize the market and provide affordable generics without the "profit-at-all-costs" mandate.
The Reality Check
The "revolving door"—where high-ranking government officials leave their posts to take high-paying jobs at the companies they used to regulate—is perhaps the clearest example of the corruption you’re highlighting. It makes it very difficult for a regulator to be "tough" when their future employer is sitting across the table.
Do you believe the fix is a total ban on private funding for oversight, or do we need a completely separate, taxpayer-funded body that has no ties to the industry at all?
The transition to a fully decoupled system—where the "referees" (regulators) have zero financial connection to the "players" (corporations)—is often cited by bioethicists as the only way to restore absolute public trust.
If science is to be purely data-driven, there are three major structural hurdles the current system would have to clear:
1. The Funding Gap
Currently, industry "user fees" account for nearly $3 billion (roughly 45-50%) of the FDA’s budget for human drug oversight. Replacing this with taxpayer money would require a massive shift in Congressional spending. Proponents argue this is a small price to pay to ensure unbiased medicine, but the political hurdle is significant.
2. The "Hidden Data" Problem
To base science strictly on data, we would need to eliminate proprietary data. Right now, companies treat their raw clinical trial data as "trade secrets."
The Reform: A mandate that any drug seeking approval must have its raw, anonymized data hosted on a government server accessible to independent scientists for re-analysis.
The Benefit: This would stop the practice of "cherry-picking" only the successful trials for publication.
3. Ending the Revolving Door
A ban on funding is one half of the coin; the other is a ban on personnel. Many argue for a "cooling-off period" (e.g., 5 to 10 years) where regulators cannot work for the industries they oversaw. This prevents the "soft corruption" where a regulator might go easy on a company today in hopes of a lucrative consulting gig tomorrow.
The Challenge of Global Science
One complication is that science is global. Even if the U.S. government bans private funding for oversight, a company could perform its research in a country with much looser rules and then bring that "tainted" data back to the U.S. for approval. This would require an international standard for data integrity that currently doesn't exist.
The Bottom Line: Shifting to a 100% publicly funded oversight model would treat healthcare as a national security or public utility issue rather than a market commodity. It would prioritize the "truth" of the data over the "speed" of the market.
If we moved to this model, do you think the public would be willing to accept slower drug approvals in exchange for knowing the data is 100% untainted?